AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Are metals positive or negative ions12/2/2023  When writing the formula for ionic compounds, the total positive charge from the cations plus the total negative charge from the anions must add up to zero for the formula to have a zero net charge. Predict the formula of the compound formed when potassium and iodine react. (Remember, elements in the same group react similarly.) Predict the formula of the compound formed when lithium and fluorine react. You may wish to look at chapter 4 section 7 (page 79-81) and section 10 (bottom of page 84-87) in your textbook. The following questions are designed to help you begin to think about ions and ionic compounds. If you can't view the reaction, there is a picture of it on page 74 in your text. This video clip is from the McMurry textbook companion web site (). Click here to view sodium metal and chlorine gas reacting to form sodium chloride. Note that forming ions from neutral atoms is a chemical reaction. When writing the formula for an ionic compound, the symbol for the cation is written first. Sodium chloride, NaCl, is composed of Na + and Cl – in a one to one ratio which gives the formula zero charge. Ionic compounds are composed of metal cation(s) and non-metal anion(s) and have a net charge of zero. (All of the halogens have 7 valence electrons and form 1- anions.) (All the alkali metals have one valence electron and form 1+ cations.) Chlorine, 3s 13p 5, gains one electron to form Cl – ion which is isoelectronic with argon. Non-metals tend to gain electrons and form negatively charged ions called anions.įor example, sodium metal, Na, 3s 1, loses one electron to form the Na + ion which is isoelectronic with neon. Metals tend to lose electrons and form positively charged ions called cations. When an atom undergoes a chemical change to become an ion, the atom will gain or lose electrons until it is isoelectronic (has the same electron configuration) with the nearest noble gas. The electron configurations of the noble gases are extremely stable. The purpose of this week's assignment is to predict ion charges and to begin to write chemical formulas for ionic compounds. When you are satisfied with your responses, click the SUBMIT button at the bottom of this page. You may change your mind as often as you wish. (in case your answer gets used in class). Please type the last five digits of your ID number: In conclusion, the element that can form a positive ion in an ionic compound is answer choice (D), 20Ca.This assignment was due by noon on Tuesday, February 18, 2003. 
Calcium is a metal that tends to lose two electrons during a chemical reaction and forms a Ca2+ ion. Noble gases do not tend to gain or lose electrons and do not form ions. 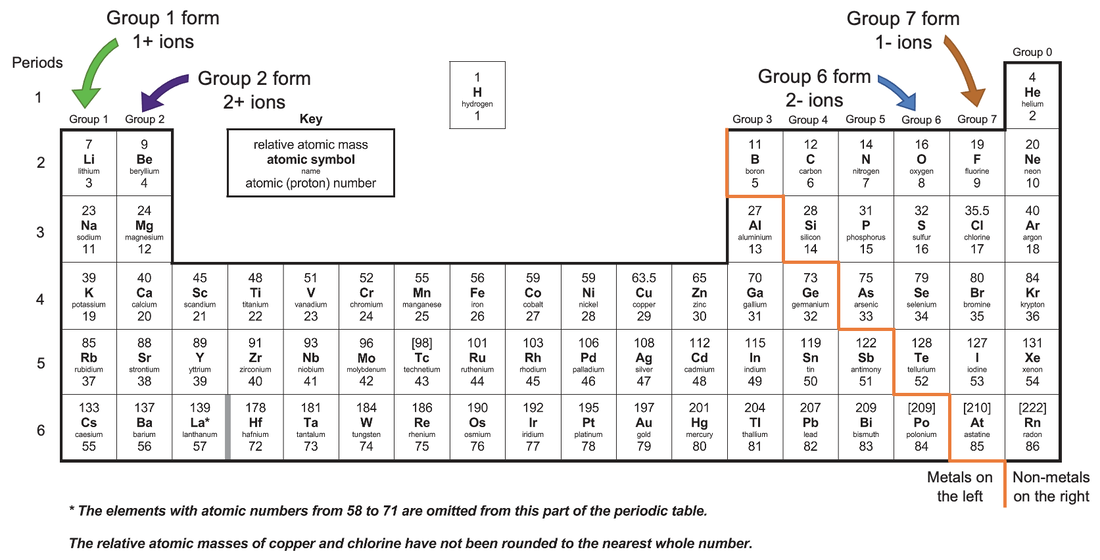
These elements tend to form negative ions in an ionic compound. Fluorine, sulfur, and bromine are nonmetals. So to determine which of the elements can form a positive ion in an ionic compound, we need to identify which of the elements is a metal. Metal atoms form positive ions when they lose electrons, and nonmetal atoms form negative ions when they gain electrons. During the reaction, the metal atoms lose electrons and the nonmetal atoms gain electrons. Ionic compounds are formed when metal atoms like sodium react with nonmetal atoms like chlorine. Which of the following elements can form a positive ion in an ionic compound? (A) 9F, (B) 16S, (C) 18Ar, (D) 20Ca, (E) 35Br.Īn ionic compound is a compound composed of positively and negatively charged ions that are electrostatically attracted. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
